Ozanimod (brand name Zeposia) is rapidly becoming a cornerstone in the treatment of relapsing forms of Multiple Sclerosis (MS). For professionals in the pharmaceutical and ADL sectors, understanding its unique “trap-and-contain” mechanism is key. Here is a concise breakdown for your LinkedIn audience. 🔬 Science Spotlight: How Ozanimod Works Unlike traditional therapies that might broadly […]

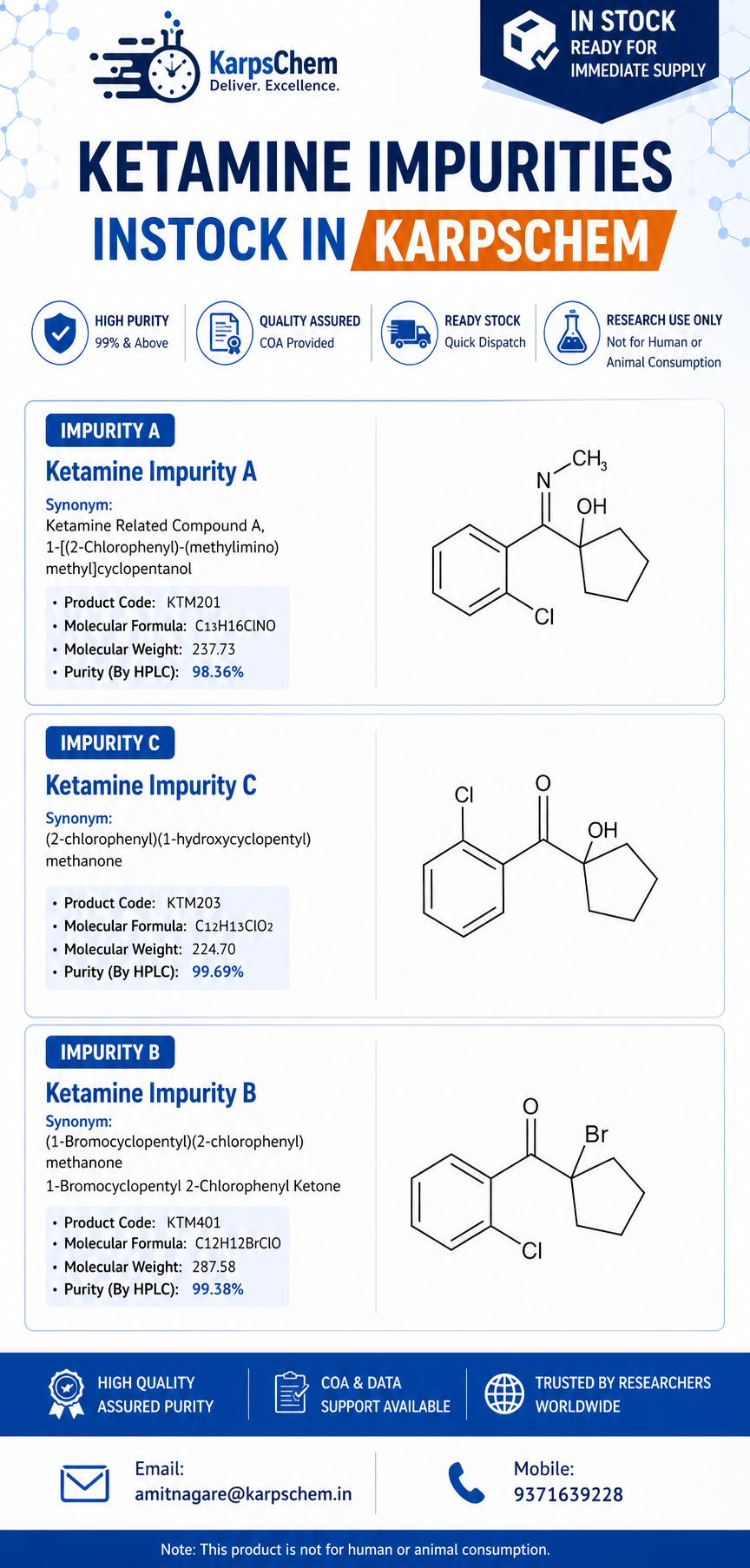
🚀 Ketamine Impurities In Stock at Karpschem Laboratories
In the fast-evolving pharmaceutical landscape, access to high-quality reference standards is critical for accurate analysis, regulatory compliance, and successful product development. At Karpschem Laboratories Pvt. Ltd., we are proud to offer a wide range of Ketamine impurities that are readily available in stock for immediate dispatch. 🧪 Available Ketamine Impurities ✔ Ketamine Impurity A✔ Ketamine […]

Teneligliptin Impurities In Stock at Karpschem Laboratories – Complete Range for Pharma R&D
In today’s competitive pharmaceutical landscape, ensuring drug purity, safety, and regulatory compliance is critical. For companies working with Teneligliptin API, access to high-quality impurity standards is essential for analytical development and regulatory submissions. Karpschem Laboratories Pvt. Ltd. is pleased to announce that a comprehensive range of Teneligliptin impurities is now in stock, ready for immediate […]

Tolterodine Impurities In Stock at Karpschem Laboratories – Trusted Source for Pharmaceutical Standards
In the rapidly evolving pharmaceutical industry, the demand for high-purity impurity standards has become more critical than ever. For companies working on Tolterodine-based formulations, sourcing reliable impurity standards is essential for regulatory compliance, analytical development, and quality control. Karpschem Laboratories Pvt. Ltd. is proud to announce that Tolterodine impurities are now in stock, available for […]

Complete Range of Brompheniramine Maleate Impurities Now Available at KarpsChem Laboratories
Understanding and controlling impurities is a fundamental requirement in pharmaceutical development. As regulatory expectations continue to rise, access to well-characterized, high-purity impurity standards becomes essential for accurate method validation, impurity profiling, and stability studies. At KarpsChem Laboratories, we are committed to supporting the pharmaceutical industry with reliable, research-grade impurity standards synthesized under strict quality systems. […]

All Glycol Impurity Derivatives Now In-Stock at KarpsChem Laboratories
At KarpsChem Laboratories, we continue to expand our high-purity impurity standards portfolio to support global pharmaceutical, chemical, and research companies. We are pleased to announce that a comprehensive range of Glycol impurities and their derivatives are now readily available in stock. Available Glycol Impurity Standards: Ethylene Glycol Monoisopropyl Ether Polyethylene Glycol 6000 (PEG 6000) Diethyl […]
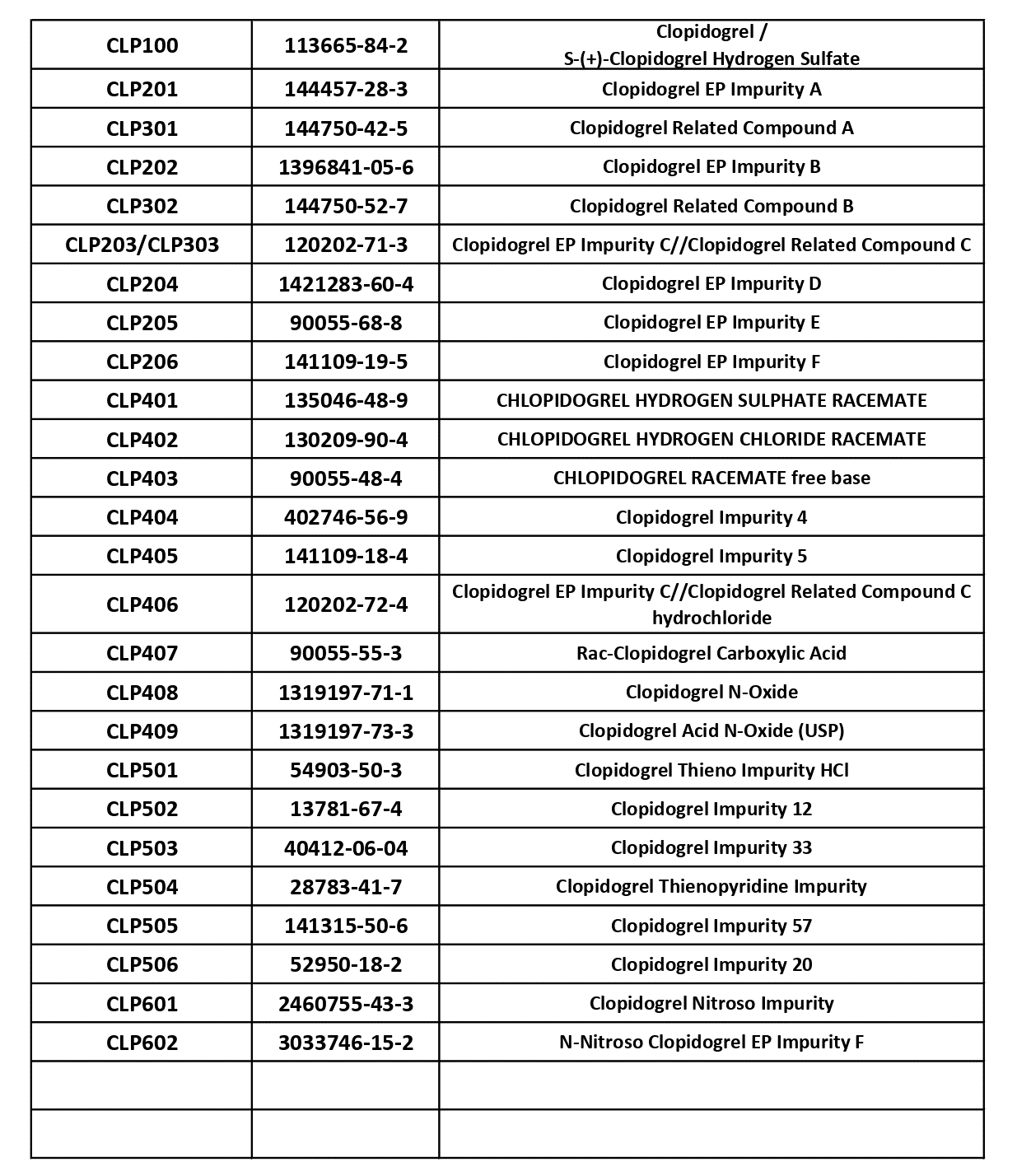
Clopidogrel Impurities – Complete Solutions Available at KarpsChem
Clopidogrel is one of the most widely used antiplatelet drugs in cardiovascular therapy, and accuracy in impurity profiling plays a critical role in ensuring its quality, safety, and regulatory compliance. As pharmaceutical companies continue to advance research and generic drug development, the demand for high-purity Clopidogrel impurities and reference standards has increased dramatically. At KarpsChem, […]
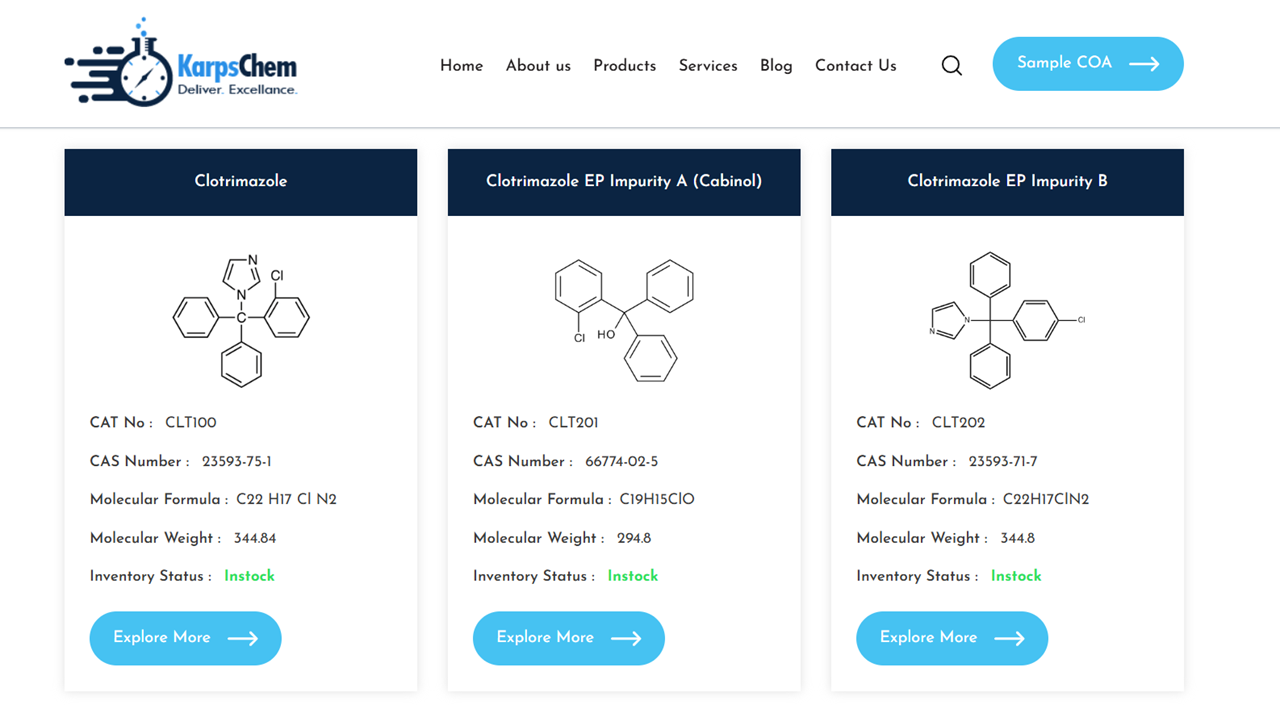
Comprehensive Guide to Clotrimazole EP Impurities (A–F) | High-Purity Standards at KarpsChem
Clotrimazole is a widely used antifungal agent, belonging to the imidazole class, primarily indicated for the treatment of dermatomycoses, candidiasis, and other fungal infections. To ensure regulatory compliance and drug safety, it is essential to identify, synthesize, and study its pharmacopoeial impurities as per EP/BP/USP/ICH guidelines. At KarpsChem Laboratories Pvt. Ltd., we specialize in providing […]

Bile Acid Impurities – High-Purity Reference Standards Available at KarpsChem
India has become a global hub for the manufacturing and supply of bile acid impurities, supporting industries in pharmaceutical research, drug development, and academic studies. Among the leading suppliers, KarpsChem stands out for its expertise in custom synthesis, impurity profiling, and high-purity reference standards. Our inventory includes a wide range of bile acid impurities with […]